Catherine Dulac, Joseph Bergan and Yoram Ben-Shaul (Courtesy of Department of Molecular and Cellular Biology)
Sex-based differences in sensory processing identified at cellular level for first time
by Parizad Bilimoria
Take a moment to picture the reaction of a man in a bar to a beautiful woman walking in the door. Then picture the reaction of a woman sitting in the same spot in the same bar, looking at the same thing. Chances are you imagined different behaviors.
“The most extreme behavioral differences within a species are found in the way male vs. female animals respond to the same sensory stimuli,” write the authors of a recent study on sensory processing in the mouse brain, led by Catherine Dulac, Higgins Professor of Molecular and Cellular Biology in the Department of Molecular and Cellular Biology at Harvard and a Howard Hughes Medical Institute Investigator.
But why? How are the brains of males and females different, and how does this lead to behavioral differences? Consumers of pop culture and science alike are dying to know. Hypotheses are explored in forums ranging from fashion magazines in the checkout aisle of the grocery store to academic journals in neuroscience, psychology and medicine. Scientists have long known about sex-based hormonal differences in the brain and are beginning to identify subtle structural differences; however, at the cellular level these questions remain a mystery.
The Dulac lab’s recent research reveals a population of neurons that “fire” or respond to the same sensory stimuli very differently in male vs. female mice. These neurons are present in the medial amygdala, a deep brain structure known for triggering instinctual behaviors—such as mating, parenting, or territorial defense—that are sexually dimorphic in mice.
“This is the first paper that shows a population of neurons that are functionally different in male and female mammals,” says Joseph Bergan, a postdoctoral fellow in Dulac’s lab who conducted the study along with co-author Yoram Ben-Shaul, now at The Hebrew University of Jerusalem. “On a cell by cell basis, there are these very profound functional differences. A given neuron in a male animal actually responds very differently to the sensory world than a matching neuron in a female animal.”
Sensory signals that matter
For mice, the sensory world Bergan alludes to is filled with the odors of other animals. Unlike humans, mice rely much more on their olfactory senses than vision. Thus much of the sensory input coming into the amygdala—which processes and sends sensory information onto the hypothalamus, another deep brain structure that governs innate behaviors and the neuroendocrine system—is from the olfactory system in mice. Specifically, in the medial amygdala most of the olfactory input comes from the vomeronasal organ, a structure in the back of the mouse’s nose that is specialized for sensing chemical cues in bodily fluids such as urine, sweat, tears or saliva.
Bergan and Ben-Shaul zoomed in on the activity patterns of medial amygdala neurons because they hoped to better understand how the brain processes sensory information and uses it to guide innate behaviors. And because they were studying mice, they focused on sensory information that matters in the survival and social lives of these animals—such as the urine of predators or other mice.
They found that medial amygdala neurons of males and females responded strongly to predator urine at all ages tested. But after puberty, the same population of deep brain neurons displayed dramatic differences in response to urine from same vs. opposite sex members of their own species. The neurons in male mice responded fervently to female urine, and in female mice to male urine.
These cellular activity results are in line with what one might predict behaviorally—that predator cues would scare off both males and females alike, young or old, while social cues from same vs. opposite sex members of one’s own species might elicit distinct responses in animals ready to mate.
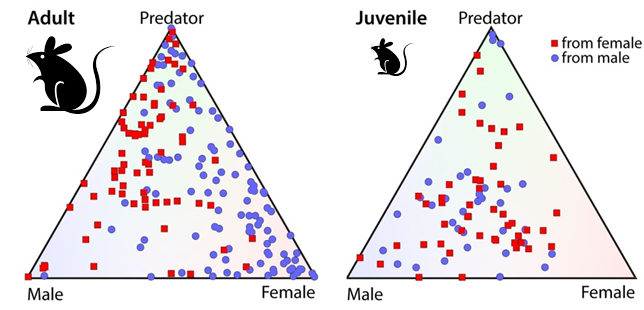
The role of sex in neural responses to odor. Each red dot represents a deep brain (medial amygdala) neuron in a female mouse, and each blue dot an analogous deep brain neuron in a male mouse. The corners of the triangles represent the different odor cues tested (predator urine, male mouse urine, and female mouse urine). The position of each dot reveals the cell’s firing pattern. A dot in the center represents a neuron that is often “turned on” by all three stimuli, whereas a dot in one corner of the triangle represents a neuron that is usually “turned on” specifically by that stimulus. Clearly, neurons from adult females respond strongly to male odor cues and neurons from adult males respond more strongly to female odor cues. Juvenile mice, on the other hand, lack this sex-based difference. (Courtesy of Dulac Lab)
But the authors didn’t stop at studying the amygdala neurons in wild type (“normal”) lab mice. Using genetic manipulation, they proceeded to see if they could tie the new results in with was already known about sexual dimorphism in the brain—namely, that levels of the sex hormone estrogen, synthesized from testosterone by an enzyme called aromatase, play a key role in differentiating male and female brains. It turns out that adult mice lacking aromatase do not exhibit clear sex-based differences in their response to same species urine. So while the discovery of the sex-specific odor processing neurons is new, their development can indeed be linked with existing knowledge on the brain’s sexual differentiation.
An elusive bunch of neurons
Before this study, Bergan says, it was long predicted that sexually dimorphic neurons controlling innate behaviors existed. The real challenge was finding them. While he and his colleagues hypothesized that the medial amygdala, with its projections to the hypothalamus would be a good place to look, for a long time they couldn’t see any neural activity in the region.
This was unusual. Both were electrophysiology experts with experience recording neural activity patterns in various brain regions, and usually they found some baseline activity to measure and build their experiments upon. But here there seemed to be virtually nothing at baseline.
Frustrated, they one day gathered all the olfactory stimuli they could find in the lab and took turns randomly presenting them to the animals while recording activity in the medial amygdala—essentially a trial and error method of attempting to ‘awaken’ the quiet neurons.
They felt a bit silly at first, Bergan recalls. “But as we started to do that, neurons started to appear from nowhere. That was actually a pretty fun day.”
After that day, their experiments finally fell into place and data began streaming in slowly but surely. Bergan is enthusiastic not just about the specific findings of the present study, but also, more broadly, the doors it has opened for future studies.
“I find it pretty exciting that we are starting to be able to record the moment-to-moment activity of neurons and map these differences onto profound differences in essential behaviors. This gives us an entry point to explore causal relationships between neural activity and specific behaviors,” he says.
The next step will be to explicitly link the differences in neural activity observed in these experiments to the known differences in behavior between males and females, adults and juveniles.
“This is an exciting time in neuroscience,” says Dulac. “We have tools that enable us to record, silence or activate specific populations of neurons throughout the brain, and start asking very mechanistic questions about which neuron types are involved in a specific social behavior, and how their activity confers behavior specificity in young or sexually mature animals, in males and in females.”
In addition to being part of the Department of Molecular and Cellular Biology and Center for Brain Science, Dulac’s lab is also a member of Conte Center at Harvard, focused on developmental origins of mental illness, and she adds that the present research is also intriguing for its potential to shed new light on brain disorders. Many common mental illnesses such as anxiety, depression, autism or ADHD, for instance, have well known differences in prevalence in men and women.
“One of the first signs of a mental disorder is the inability to engage into proper social interactions,” she notes. “It is only when we understand the identity and mode of functioning of social behavior circuits, and how they differ according to the sex and physiological status of individuals, that we will be able to significantly improve upon the diagnosis and treatment of specific mental illnesses.”